 |
This item is on a 31 business day lead time. If you need something available sooner, click here for the GenBody COVID-19 Rapid Antigen Test Kit (25/Box)
This item can only ship to a medical or educational facility.
FDA Emergency Use Authorization
The QuickVue SARS Antigen Test detects SARS-CoV-2 antigens directly from anterior nares swab specimens. The QuickVue SARS Antigen Test provides accurate and reliable results in 10 minutes, allowing for testing of patients suspected of COVID-19/2019-nCoV in near-patient testing environments.
Features and Benefits
- Dipstick Test Format: Simple workflow follows a similar format to CLIA-waived QuickVue assays. No instrument necessary.
- Results in 10 Minutes: Test and treat in the same office visit.\
- Two-Color Result: Easy to read and interpret. Blue control line and red test line.
- All Components Included in Kit: Ready to use, no need for additional equipment.
- Room Temperature Storage: No refrigerator space needed. No need to wait for reagents to warm up. Rapid SARS-CoV-2 tests can be run immediately as needed.
Test Procedure
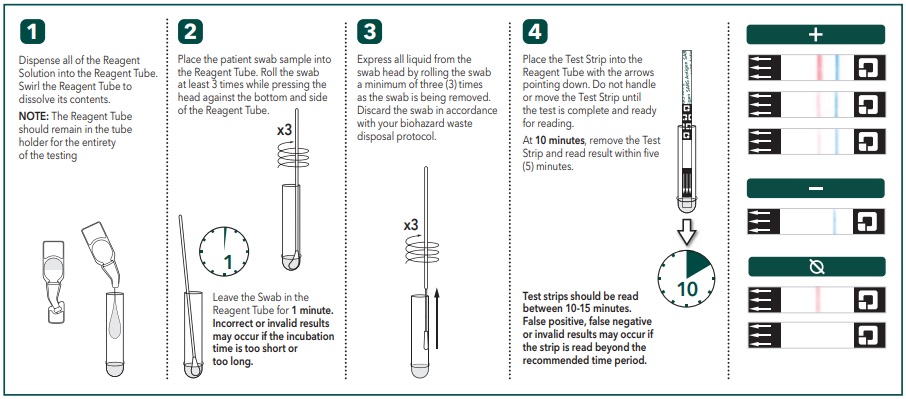
Materials Included
- (25) Individually Packaged Test Strips: Monoclonal anti-SARS antibodies
- (25) Reagent Tubes: Lyophilized buffer with detergents and reducing agents
- (25) Reagent Solution : Vials with 340 μL salt solution
- (25) Sterile Nasal Swabs
- (1) SARS Positive Control Swab: Swab is coated with non-infectious recombinant SARS antigens
- (1) Negative Control Swab: Swab is coated with heat-inactivated, non-infectious Streptococcus C antigen
- (1) Package Insert
- (1) Procedure Card
Technical Specifications
- Sample Type: Anterior nares swab
- Time to Results: 10 minutes
- Kit Storage Conditions: Room temperature (15°C to 30°C/59°F to 86°F)
- Internal Controls: Included
- External Controls: Positive and negative included
- PPA (Reference Extracted SARS-CoV-2 RT-PCR Assay): 96.6%
- NPA (Reference Extracted SARS-CoV-2 RT-PCR Assay): 99.3%
- Shelf Life: 12 months from date of manufacture
|
|
 |


 QuickVue SARS Antigen Test
QuickVue SARS Antigen Test 


