 |
This product has been discontinued. For comparable products, click here for the following:
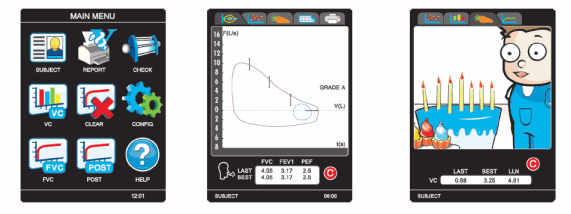
Performing spirometry tests, in any environment, could not be easier
than with the new Micro Direct In2itive spirometer. Simply enter the
patients details on the color touch-screen display, select FVC or VC
and begin the test series.
Test information is presented in an easy to read format on a clear,
bright color display. The best test is automatically selected and
built-in quality control checks insure accurate results. Test data
along with the diagnostic interpretation and estimated lung age can be
instantly printed via an external printer or downloaded to the included
Spirotrac PC software for permanent storage and trend analysis.
The in2itive features a highly accurate Fleisch pneumotach flow
measuring system, battery operated portability and is shipped complete
with all necessary accessories in a robust carrying case. The easy to use and affordable In2itive spirometer provides a fast, accurate and cost effective spirometry solution.
Features and Benefits
- Rapid, low cost testing
- Robust Fleisch pneumotach
- 8 1/2" x 11" printout via USB printer or PC
- Rechargeable battery pack
- Real-time FV or VT curves
- Choice of child incentives
- Simple icon driven menus
- On-screen test quality prompts
- Pre/Post bronchodilator comparison
Spirotrac V Software Features
- Intuitive user interface with workflow-orientated presentation
- Subject list, test session list, dynamic information panel and session data in one view
- Automatic FEV1 trend chart view as subject is selected
- Open session, f/v & v/t curves, all test data, test quality and subject demographics in one view
- Automatic storage of all test data and events, including calibration log
- Attractive incentive displays for children, designed to maximise interest and effort
- View real-time curves through the incentives
- Comprehensive reports for spirometry and trending including PDF generator for remote electronic filling and e-mailing
- Configurable spirometry reports
- Manual data entry and data export facility
- Mannitol/Bronchialitol/Bronchial Challenge Testing
Materials Included
- Hand-held Spirometer
- USB cradle
- Spirotrac V PC Software
- Protective Carrying Pouch
- User Manual
Screen Shots
Technical Specifications
- Subjects: Automatic population of database from Spirotrac. Name, ID, Age, Height, Gender, Smoking Status, Body Mass Index and more depending on model. Storage of up to 10,000 subjects
- Environmental Data: Selectable; Temperature (built in sensors), Barometric Pressure, Altitude, Humidity
- Test Types: Single breath tests, flow/volume loops, multi-breath testing, tidal breathing and combined VC/FVC type test methods supported
- Parameters Measured (depending on model): VC, IVC, IC, VT (TV), TLC, RV, IRV, ERV, FRC, FVC, FIVC, FEV1, FEV3, FEV6, FEV1/VC, FEV1/FVC, FEV3/VC, FEV3/FVC, EV1/FEV6, FEF75, FEF50, FEF25, FEF25-75, FEF25-75/FVC, FIV1, PIF, FIV1/FIVC, FIF25, FIF50, FIF75, FEF50/FIF50, FET, MVVind, FEV1 Ratio, FEV0.5, PEF L/min, PEF L/sec, FEF 0.2-1.2, FEF75-85%, FEF25%, FEF50%, FEF75%, FMFT, FIF25%, FIF50%, FIF75%, PIF, FEV0.75, Lung Age and more
- Predicted Normals: NHANES III, Polgar, Knudson, Crapo, Hsu and many more
- Suggested Interpretation: User Selectable
- Flow Technology: Fleisch Pneumotachograph
- Resolution: 10 mL volume; 0.01 L/s flow
- Data Storage: Stores up to 10,000 subjects and hundreds of test sessions
- Accuracy when in Operating Range:
- Volumes: Better than +/- 3% (0 - 10 L)
- Flows: Better than +/- 10% (0.02 - 16 L/sec)
- Linearity: +/- 5% in range 0.1 to 16 L/s
- PowerSAFETM:
- Input 100 - 240V AC 50-60Hz
- Output - 5V DC
- Battery Pack: Rechargeable Lithium Polymer
- Dimensions: 6 1/4 x 4 x 1 3/4 with flowhead attached
- Storage Humidity: 10% to 95%
- Storage Temperature: 32-122oF
- Recommended Operating Range: 63-98oF; Design Limits: 50 - 104oF
- Ambient Humidity: 0% to 99%
- Connectivity: USB 2
- Max Test Duration: 20 sec. FVC; 30 sec. VC
- Performance Standards: ATS/ERS 2005
- Safety Standards: IEC 60601-1:2005
- Medical Safety Standards: Medical Devices Directive 2007/47/EC
- Designed & Manufactured Under: ISO 13485:2003, FDA 21CFR820 CMDR & JPAL
Warranty Information
|
|
 |
